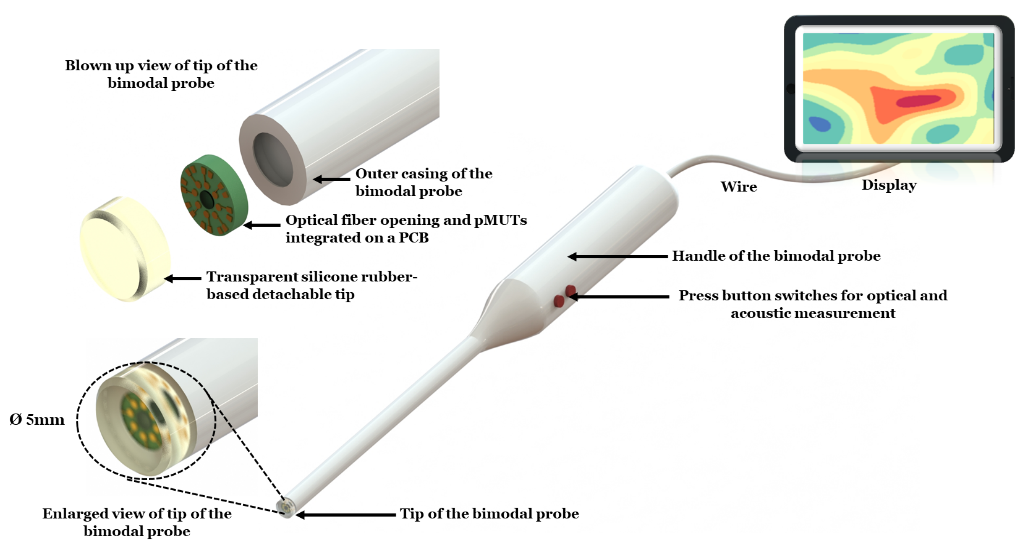
Development and clinical validation of a multimodal intraoperative probe for breast cancer margin assessment.
Problem Statement:
Breast cancer occurs in 1 out of 22 women in India. The early and proper pathological diagnosis leads to definite treatment. The Gold standard procedure is to delineate the tumour boundaries is histopathology after the resection of the tumour at the time of surgery. It takes a couple of hours to get the result, which increases the surgery times, or the patient is discharged by that time. if the false positive margin is detected, then the chances of reoccurrence increase drastically.
Concept:
The project proposes to develop a tool that can detect changes in the optical properties of biopsied tissues as cancer progresses and hence use these signatures for a possible early diagnosis of different types of cancer. The technique used near-infrared absorption spectroscopy to analyse the optical properties of the biopsied tissue. In addition to the optical modality, the proposed probe will include the acoustic modality where ultrasound imaging can define tumour boundaries. The difference in density, stiffness and elasticity of cancerous tissues with respect to normal tissues will define the boundaries in Ultrasound imaging.
Technology/Science involved:
MEMS sensots, Ultrasound imaging, pMUTs, Breast physiology
Design and Development of Optical Modality-based Probe for Breast cancer Diagnosis
Problem Statement:
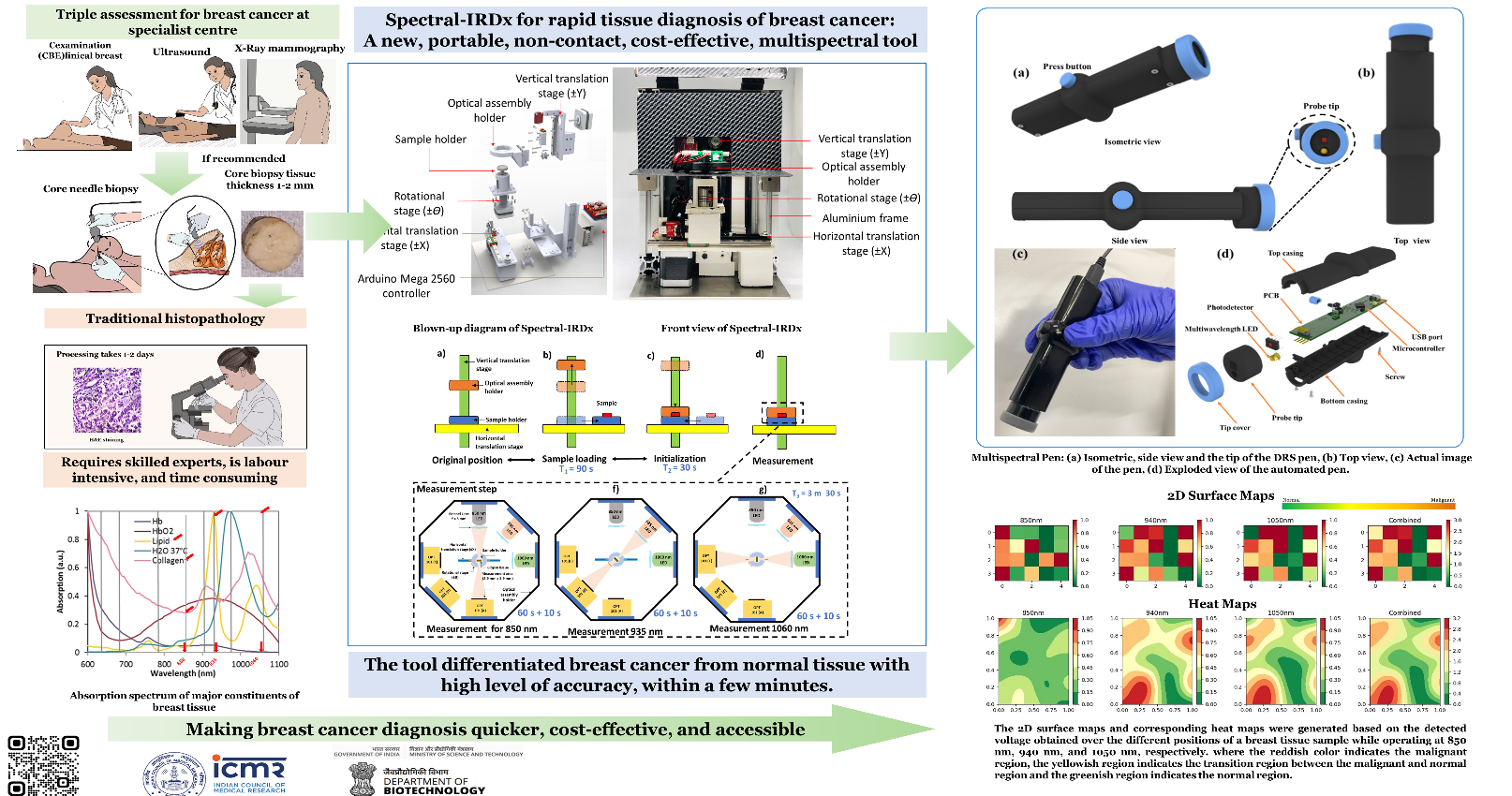
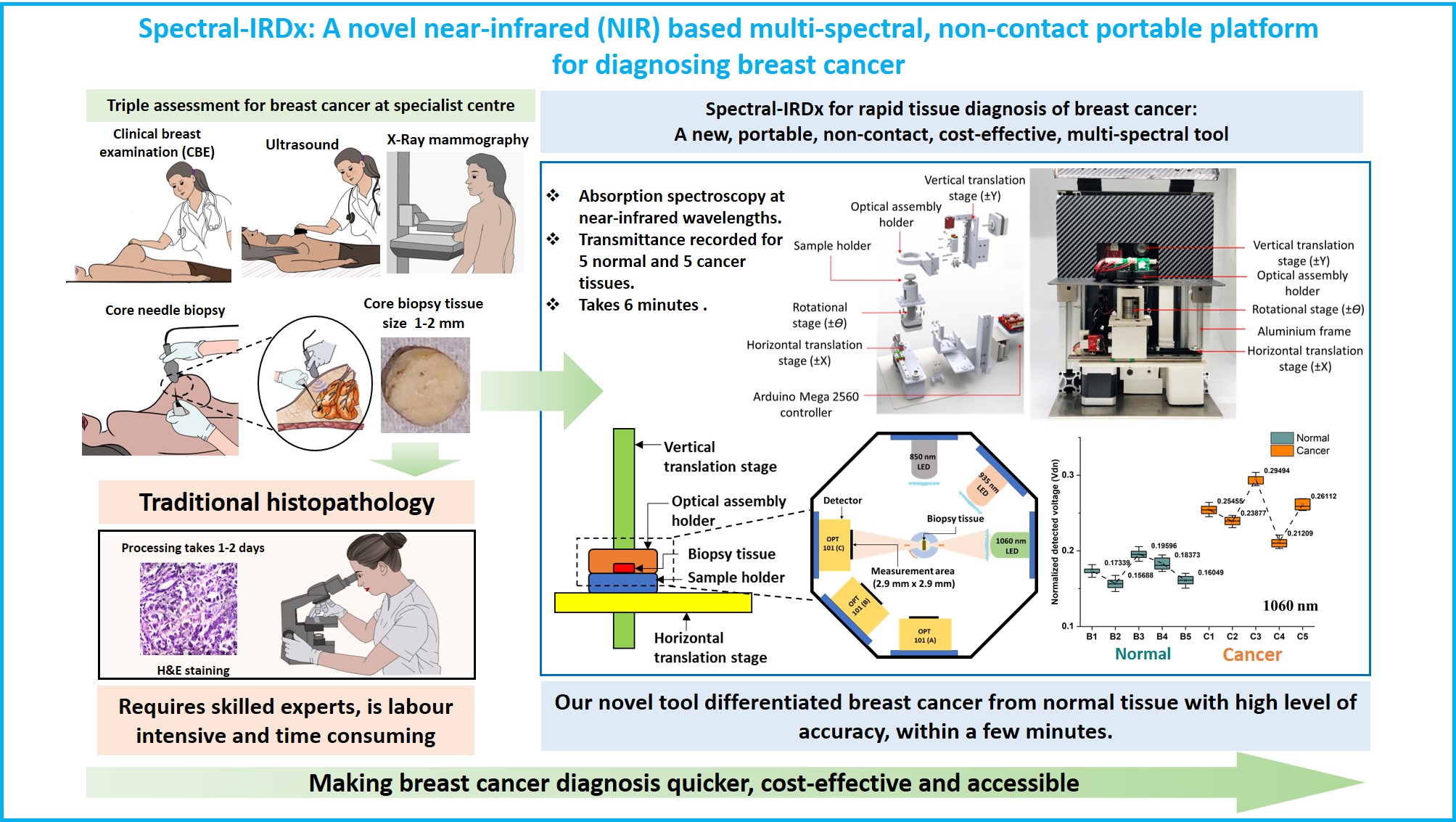
According to the International Agency for Research on Cancer (IARC), breast cancer is one of the most widespread forms of cancer, affecting around 2.3 million women annually and contributing to nearly 7% of mortality among all cancers found in women. Breast cancer ranks first and accounts for around 11.7% of the commonly diagnosed cancer cases as per IARC in 2020. The histopathology technique (gold standard) involves freezing the excised breast tissue and slicing the tissue by hundreds of micrometers. This sliced tissue is further stained using hematoxylin and eosin (H&E) and investigated using a microscope by the pathologist. A core needle biopsy or fine-needle aspiration cytology (FNAC) may also be performed to ascertain the biochemical nature (mainly Estrogen, Progesterone, and HER2 status) of the malignancy. Often breast-conserving surgery, i.e., lumpectomy or local excision of diseased tissue, is done under the guidance of an ultrasound probe, known as a minimally invasive technique for resection of diseased tissue. The main aim of minimally invasive surgery is to achieve complete removal of the tumor while leaving normal breast tissue intact with a better cosmetic outcome, less patient anxiety, decreased morbidity, and reduced economic burden to the patients. Intraoperative cancer margin assessment plays an essential role in ensuring the success of the surgery. In this direction, the developed multispectral diffuse reflectance spectroscopy (DRS)-based intraoperative handheld probe (Multispectral-Pen) can play a vital role in characterizing malignant tissues from adjacent normal tissues to assess the tumor margin.
Concept:
The project involves the design and development of a probe (i.e., Multispectral Pen) that can help in the delineation of adjacent normal and cancerous regions intraoperatively using the diffuse reflectance spectroscopy technique based on the scattering and absorption behavior of the tissue. Multispectral Pen can aid the surgeon during a lumpectomy surgery to remove cancerous tissue while keeping adjacent normal tissue intact. The surgical resection can be pre-planned utilizing pre-operative imaging (e.g., X-ray mammography, MRI, or Ultrasonography) to get the approximate location of the cancerous region. A Graphical User Interface (GUI) is developed for an interfacing module (i.e., laptop or tablet) to control the Pen to acquire the data and process the data for a more straightforward interpretation.
Technology/Science involved:
Near-infrared Spectroscopy, Breast Cancer, Label-free, Optical phenotyping, Breast Anatomy, Diffuse Reflectance Spectroscopy
Electro-thermo-mechanical phenotyping of breast cancer for aiding pre-operative staging
Problem Statement:
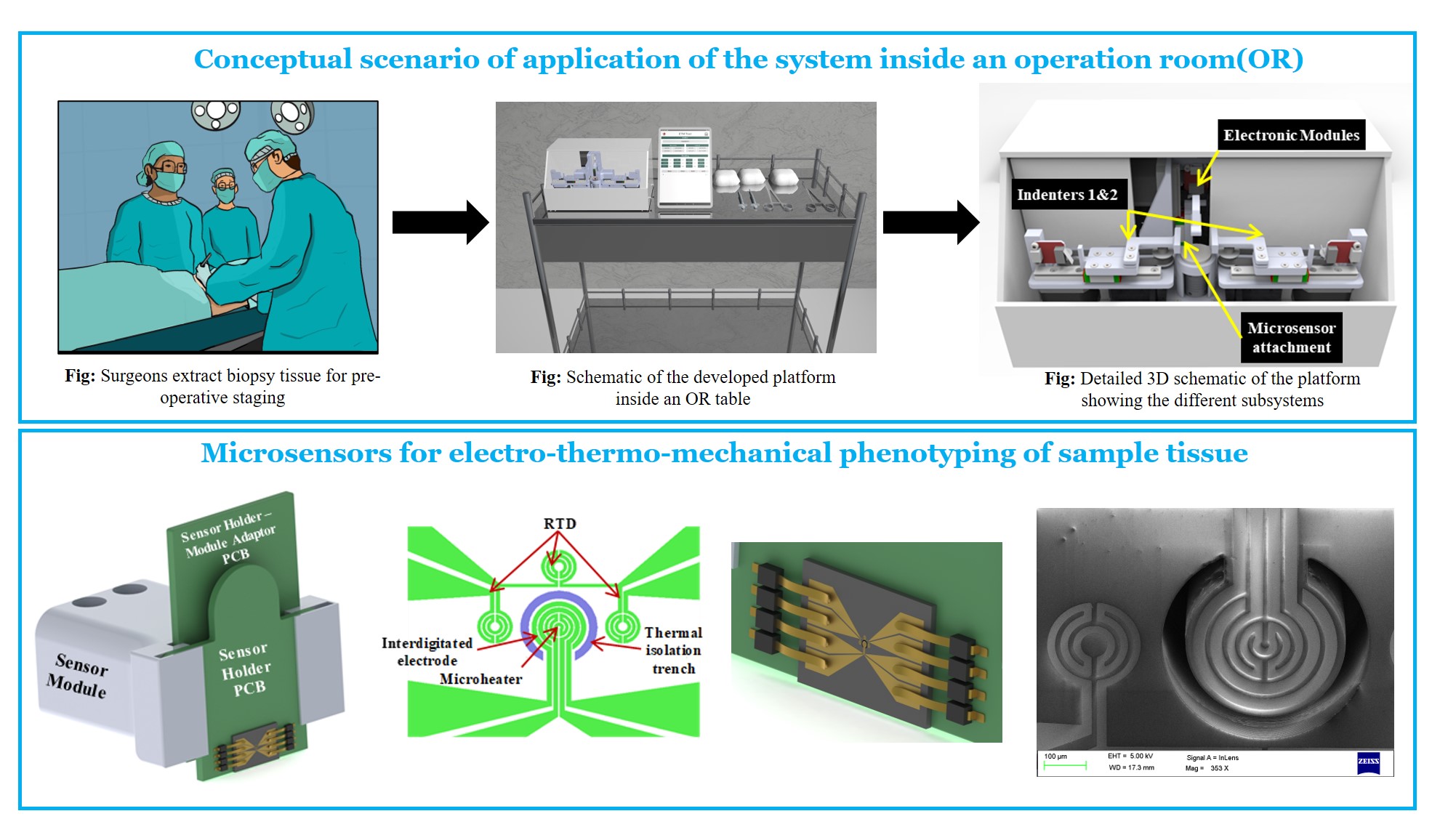
Breast cancer is the most commonly occurring cancer in women with over 2 million new cases reported in 2018. The gold standard diagnostic techniques for breast cancer are immunohistochemistry and histopathology which look for the key biomarkers of breast cancer and atypical morphology of cells. These techniques have limited use inside the operation room (OR) for pre-operative staging during surgery owing to time constraints. The existing standard method for determining margins during breast cancer surgery is frozen section examination which requires the surgically removed sample to be sent to the lab. There are no commercially available tools which can be used inside the operation theatre to perform rapid phenotyping of core biopsy. The research problem attempts to leverage the difference in physical properties between cancerous and normal tissue to aid in pre-operative staging inside the OR.
Concept:
A portable platform is developed that is capable of probing and characterizing the physical properties of a cancer biopsy tissue such as its electrical impedivity, thermal conductivity, and mechanical stiffness. MEMS-based sensors are attached in a modular fashion to the probing indenters of the system and the on-board electronics measures and computes the various physical characteristics of the sample tissue. Based on the computed parameters, the system then determines whether the tissue under testing is cancerous or not. This information is then used to assess the margin to be kept for resection during breast cancer surgery.
Technology/Science involved:
Molecular oncology, MEMS sensors, mechatronics, Rapid-prototyping, Biophysics, Physical oncology, Mammary gland biology, Breast physiology
Patent Application number:
Design and Development of Optic-based Diagnostic Tool for Breast Cancer
Problem Statement:
Breast cancer is the most common cancer, reporting a massive 2,088,849 new cases (11.6% amongst all cancers) and 626,679 deaths (6.6% amongst all cancers) worldwide in 2018. Current methods for diagnosis of breast cancer, whether in a symptomatic or a screening situation, are clinical breast examination, imaging with X-ray mammography, and ultrasound as well as tissue diagnosis using needle biopsy. The most common tool used for screening of breast cancer is X-ray mammography, which has been reported to have sensitivity and specificity of 77% and 97% respectively. However, X-Ray mammography’s sensitivity and specificity reduce significantly to 67% and 89% for dense breasts. Moreover, X-Ray mammography is expensive, hospital-based, involves radiation risks, and its efficacy in patients less than 40 years is not yet established. Contrast-enhanced Magnetic Resonance Imaging (MRI) is reported to have the highest sensitivity between 93% and 100%; but is costly, bulky, and hospital-based. While ultrasonography requires a skilled operator and gives high false positives compared to X-ray mammography. Thermography is highly sensitive to ambient and temperature fluctuation, resulting in high false-positive rates, low accuracy, and is not used in clinical practice. Additionally, women living in limited-resource settings, and geographically remote areas do not have easy access to high-quality diagnostic facilities traditionally designed for secondary or tertiary referral centres. Furthermore, with rising healthcare costs, even in the developed world, there is a need for a diagnostic technique that is robust, cost-effective, and easy to interpret.
Concept:
The project involves the design, simulation, and fabrication of the optical tool (Spectral-IRDx) that can delineate between the normal and cancer biopsy tissue based on near-infrared absorption spectroscopy, ultrasound spectroscopy, and photo-acoustic technique. For validation of the study, the spectroscopy and photo-acoustic data are compared with the immunohistology test. The Spectral-IRDx tool could potentially aid the onco-pathologist to make an informed decision to delineate between normal and cancer cases. The second phase of the project involves in the design and develop a real-time in-vivo intra-operative imaging tool for the assessment of tumour margin. The image quantifies the different cancer biomarkers such as lipid, collagen, haemoglobin, and water.
Technology/Science involved:
Molecular oncology, Optical tomograghy, Mechatronics, Rapid-prototyping, Biophysics, Physical oncology, Mammary Gland Biology, Breast physiology
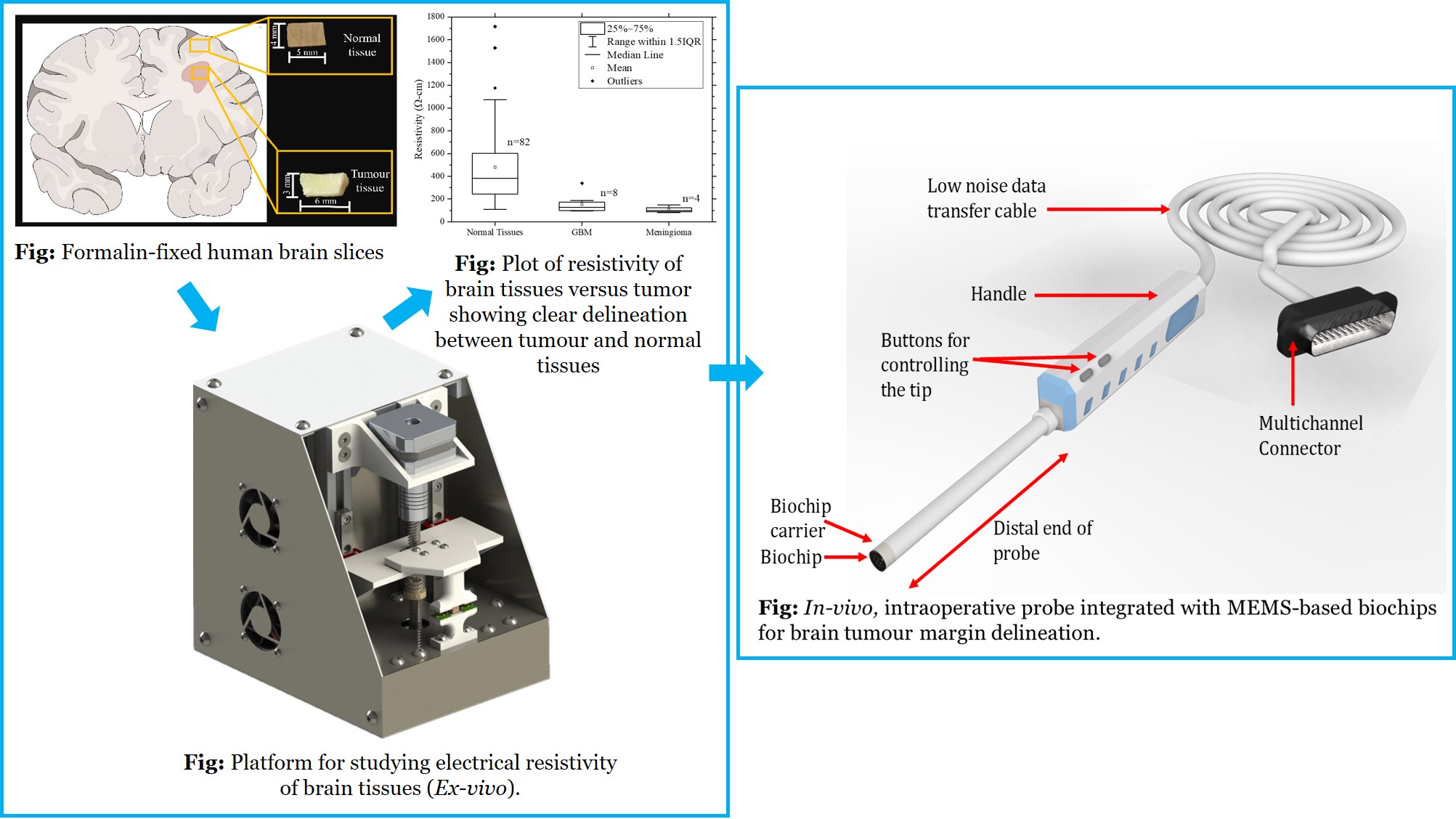
In-vivo, intraoperative probe integrated with MEMS-based sensors for improving glioma resection
Problem Statement:
Gliomas are the most common type of primary brain tumours. Despite some improvements in survival rates of glioma patient prognosis is still relatively poor. The median survival rates are in the range of 9-12 months and 2-year survival rates are in the range of 8%-12%. Surgically removing the tumour (resection), followed by radiation and chemotherapy is the usual treatment for diffuse glioma. The accuracy and efficacy of resection strongly influences the effectiveness of chemotherapy and radiation, whilst minimizing damage to surrounding tissues and associated neurological loss. Tools that can improve resection quality are thus important for the effective treatment of gliomas.
Concept:
MEMS-based sensors are fabricated using microfabrication technologies. Sensors will then be integrated to a portable platform to measure the electromechanical properties of human brain tissues (ex-vivo). The results will then be used to develop an intra-operative probe for delineating tumour region and distinguish tumour boundary from normal to enhance resection efficacy.
Technology/Science involved:
MEMS, Mechatronic systems, Neurophysiology, Neuro Oncology, Neurosurgical tools